Top 20 accounts for more than 75%! Take a closer look at the leading varieties in the 100 billion level cardiovascular and cerebrovascular drug market
Classification: Industry trends
Release time:2017-10-09
September 24 is World Heart Day. The eternal theme of World Heart Day is "Healthy Heart, Happy Life". As we all know, the heart is the only organ that cannot rest for a moment. Coronary heart disease, arrhythmia, heart failure, rheumatic heart disease, cardiomyopathy and other cardiovascular diseases seriously threaten human health, while hypertension, hyperlipidemia, hyperglycemia are the main inducements leading to stroke, myocardial infarction and peripheral vascular disease.
About 17.5 million people die of heart disease every year in the world. Although there are at least 20 million survivors of heart attack and stroke every year, most of them are at risk of sequelae and disease recurrence caused by stroke. Rigid demand drives the growth of cardiovascular drug consumption. IMS data shows that in 2016, the global cardiovascular drug market reached 89.872 billion US dollars, accounting for 8.9% of the world's total drug use.
The 2016 China Cardiovascular Disease Report shows that in 2015, cardiovascular disease deaths accounted for the first place in the total causes of death of urban and rural residents. The number of people suffering from cardiovascular diseases in China has reached 290 million, and about 2.6 million people die of cardiovascular and cerebrovascular diseases every year. The incidence of heart disease in China is high.
High concentration of cardiovascular and cerebrovascular drug market
According to the 2017 Blue Book of China's Pharmaceutical Market Development released by the CFDA Southern Pharmaceutical Economics Research Institute, in 2016, the sales of drugs in six major markets, including urban public hospitals, county-level public hospitals, urban community health care, township hospitals, physical pharmacies and online stores, were 1497.5 billion yuan, an increase of 8.3% year on year. Among them, cardiovascular, cerebrovascular, tumor immune and infectious drugs have become the frontier category.
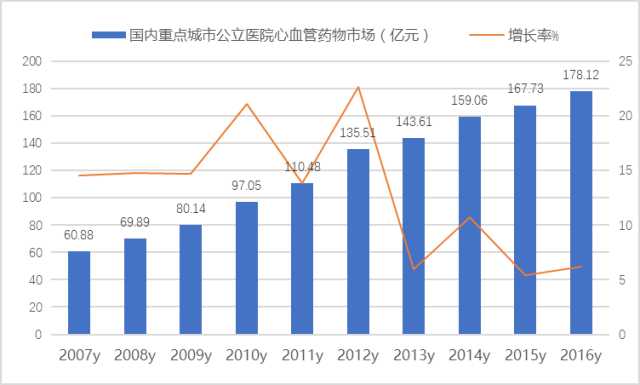
According to the HDM system data of the Southern Institute of Pharmaceutical Economics, the cardiovascular and cerebrovascular drug market in public hospitals in 16 key cities in China reached 17.812 billion yuan in 2016, an increase of 6.19% year on year. This category covers drugs for heart disease, drugs for cerebrovascular system and anticoagulant and antithrombotic drugs. Although the growth rate of cardiovascular and cerebrovascular market slowed down in recent two years, the terminal consumption of drugs showed a continuous upward trend. Data shows that the market size of cardiovascular systemized drugs, biological products and Chinese patent medicines in China has maintained a trend of continuous growth, reaching 276.9 billion yuan in 2016, an increase of 8.08% over the previous year.
Figure 1 Cardiovascular and cerebrovascular drug market of public hospitals in key cities in China from 2007 to 2016
About 17.5 million people die of heart disease every year in the world. Although there are at least 20 million survivors of heart attack and stroke every year, most of them are at risk of sequelae and disease recurrence caused by stroke. Rigid demand drives the growth of cardiovascular drug consumption. IMS data shows that in 2016, the global cardiovascular drug market reached 89.872 billion US dollars, accounting for 8.9% of the world's total drug use.
The 2016 China Cardiovascular Disease Report shows that in 2015, cardiovascular disease deaths accounted for the first place in the total causes of death of urban and rural residents. The number of people suffering from cardiovascular diseases in China has reached 290 million, and about 2.6 million people die of cardiovascular and cerebrovascular diseases every year. The incidence of heart disease in China is high.
High concentration of cardiovascular and cerebrovascular drug market
According to the 2017 Blue Book of China's Pharmaceutical Market Development released by the CFDA Southern Pharmaceutical Economics Research Institute, in 2016, the sales of drugs in six major markets, including urban public hospitals, county-level public hospitals, urban community health care, township hospitals, physical pharmacies and online stores, were 1497.5 billion yuan, an increase of 8.3% year on year. Among them, cardiovascular, cerebrovascular, tumor immune and infectious drugs have become the frontier category.
According to the HDM system data of the Southern Institute of Pharmaceutical Economics, the cardiovascular and cerebrovascular drug market in public hospitals in 16 key cities in China reached 17.812 billion yuan in 2016, an increase of 6.19% year on year. This category covers drugs for heart disease, drugs for cerebrovascular system and anticoagulant and antithrombotic drugs. Although the growth rate of cardiovascular and cerebrovascular market slowed down in recent two years, the terminal consumption of drugs showed a continuous upward trend. Data shows that the market size of cardiovascular systemized drugs, biological products and Chinese patent medicines in China has maintained a trend of continuous growth, reaching 276.9 billion yuan in 2016, an increase of 8.08% over the previous year.
Figure 1 Cardiovascular and cerebrovascular drug market of public hospitals in key cities in China from 2007 to 2016
In 2016, the amount of drugs used in public hospitals in 16 key cities for the top 20 cardiovascular and cerebrovascular products reached 13.531 billion yuan, accounting for more than 3/4 of the total, showing a high degree of product concentration. The ranking of cardiovascular and cerebrovascular TOP 20 varieties in 2016 has also changed from the previous year, and their varieties and growth trend are shown in Figure 2, Figure 3 and Figure 4. The top 10 drugs are clopidogrel, alprostadil, atorvastatin, creatine phosphate, complex coenzyme, valsartan and compound, edaravone, low molecular weight heparin, rosuvastatin and butylphthalide.
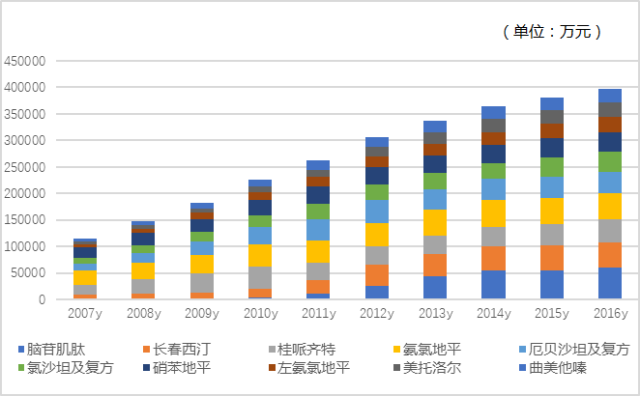
Figure 2 Overall Market of Cardiovascular and Cerebrovascular Top 20 Drugs in Public Hospitals in Key Cities in China from 2007 to 2016
Figure 2 Overall Market of Cardiovascular and Cerebrovascular Top 20 Drugs in Public Hospitals in Key Cities in China from 2007 to 2016
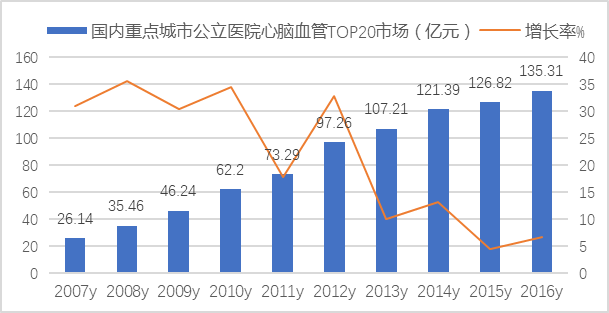
Figure 3 Top 10 drug market performance of cardiovascular and cerebrovascular drugs in public hospitals in key cities in China from 2007 to 2016
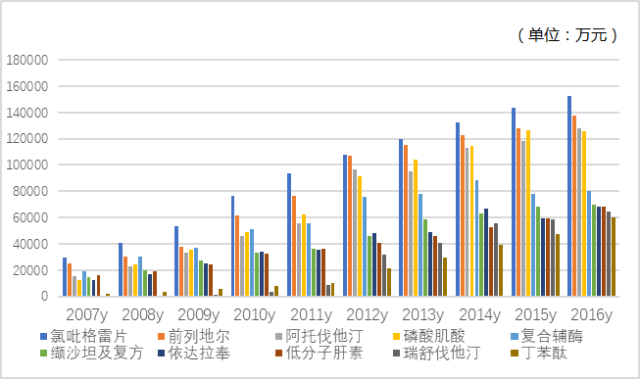
Figure 4 The market performance of cardiovascular and cerebrovascular drugs No.11-20 in public hospitals in key cities in China from 2007 to 2016
Atorvastatin leads the lipid-lowering market
The China Cardiovascular Disease Report 2016, compiled and released by the National Cardiovascular Disease Center, is the 11th annual report issued by China, which reflects the high attention paid by the Chinese government to the rising trend of cardiovascular disease. It has become an important project in the era of great health to prevent and treat hypertension, dyslipidemia, diabetes and obesity syndrome with a positive attitude of the whole people, and to take active and effective measures to curb the growth of cardiovascular disease.
The management of hypertension has a history of more than half a century, and relatively speaking, effective management of hyperlipidemia is only a nationwide action in the past 10 years. Statins have become milestones in the cholesterol lowering and blood lipid regulating program.
At present, statins in the blood lipid lowering market mainly include atorvastatin, rosuvastatin, pivastatin, simvastatin, pravastatin, fluvastatin, lovastatin, and more than 10 varieties of compound preparations, such as simvastatin+ezetimibe, atorvastatin+amlodipine, lovastatin+niacin.
Atorvastatin is the first drug in the blood lipid lowering market, and also the top three drugs in the cardiovascular market. According to CMH data of China Medical Information System, atorvastatin shows a rapid growth trend. In 2016, the sales of atorvastatin in domestic urban public hospitals were 6.075 billion yuan, the domestic retail pharmacy atorvastatin market had reached 2.431 billion yuan, and the two major mainstream terminals reached 8.506 billion yuan. The sales of atorvastatin in the six major markets of domestic urban public hospitals, county-level public hospitals, urban community medical care, township hospitals, physical pharmacies and online stores have exceeded 13 billion yuan.
According to HDM system data, in 2016, atorvastatin, rosuvastatin and pivastatin in public hospitals in key cities were among the top 3 drugs in the blood lipid lowering market, with year-on-year growth of 8.78%, 10.91% and 50.37% respectively. The six markets of the three major domestic terminals reached 23.827 billion yuan, and it is expected to reach 25 billion yuan in 2017.
Clopidogrel tops the market in heart medicine
Betrexaban, a new drug for cardiovascular and cerebrovascular systems, is another oral factor Xa inhibitor anticoagulant approved by Portola to be listed in the United States in the first half of 2017, and is the fourth drug with the same mechanism to be listed globally after rivalsaban, apixaban, and eduxaban. Betrexaban has the advantage of being an alternative to warfarin sodium, providing a new option for anticoagulation treatment of atrial fibrillation.
In the field of anticoagulant therapy for atrial fibrillation, with the introduction of rivalsaban, apixaban, dabigatran ester, tegrenol and prasugrel into clinical practice, the market for antithrombotic and anticoagulant therapy has been further improved. However, clopidogrel and low-molecular-weight heparin, which are mainstays in the Chinese market, still play an immeasurable role.
At the end of the 1990s, the US FDA approved Clopidogrel developed by Bristol Myers Squibb (BMS) to prevent and treat atherosclerotic diseases, myocardial infarction, stroke, peripheral vascular diseases and other diseases, becoming a landmark variety in the field of cardiovascular medicine.
Clopidogrel and aspirin small dose enteric coated tablets are known as the gold standard treatment plan for anti platelet aggregation and prevention of thrombosis, and are drugs to prevent myocardial infarction and stroke in the treatment of hypertension. Clopidogrel is jointly sold by Bristol Myers Squibb and Sanofi Aventis under the trade name Plavix. Clopidogrel is one of the earliest varieties developed and marketed in China. At present, 16 Clopidogrel API production lines have been built in China. In 2000, China first approved the registration and production of clopidogrel by Shenzhen Xinlitai Pharmaceutical, with the trade name of "Taijia".
Clopidogrel from Sanofi Aventis is marketed as "PLAVIX" in China. Driven by the original research drug and domestic drug, Clopidogrel has become a rapidly growing variety. The compound annual growth rate was 29.8% in the 10 years from 2005 to 2015. After the market developed to the platform stage, the growth slowed down in the past three years, and the consumption continued to rise.
HDM system data shows that in 2016, the market of clopidogrel in public hospitals in 16 key cities in China was 1.529 billion yuan, up 6.25% year on year. Among them, Sanofi Aventis's PLAVIX accounts for 58.84%, Shenzhen Xinlitai's Taijia accounts for 35.27%, and Lepu Pharmaceutical's Shuaike accounts for 5.89%. The growth rates of PLAVIX and TEGA were 5.24% and 3.66%, and the growth rate of Shueke of Lepu Pharmaceutical was 40.30%. In 2016, the overall sales scale of clopidogrel tablets in China exceeded 10 billion yuan, ranking first in the domestic heart drug market.
Figure 5 Clopidogrel market of public hospitals in key cities in China from 2007 to 2016
7 Antihypertensive drugs rank among the top 20 cardiovascular and cerebrovascular drugs
Calcium antagonists, renin angiotensin receptor antagonists and β Receptor blockers are a tripod of drugs in the antihypertensive market in China. Its mechanism of action is not only to control blood pressure, but also to protect the heart, brain, kidney and other important organs of hypertensive patients. In clinical treatment, it has positive effects on coronary heart disease, heart failure, arrhythmia, protection of renal function, reversal of left ventricular hypertrophy and vascular remodeling.
CMH data shows that in 2016, the market size of antihypertensive drugs in urban public hospitals in China was 36.6 billion yuan, an increase of 13% over the previous year. With the improvement of the medical security system, the market pattern shows that the top five calcium antagonists account for 34%, and renin angiotensin receptor antagonists account for 28%, β Blockers accounted for 8%, compound preparations for 7%, diuretics for 2%; Others accounted for 21%.
Among the top 20 varieties in the cardiovascular and cerebrovascular drug market of public hospitals in key cities, 7 drugs, including antihypertensive drugs amlodipine, levamlodipine, nifedipine, valsartan and compound, irbesartan and compound, losartan and compound, metoprolol, accounted for 21.40%.
Figure 6 Market performance of cardiovascular and cerebrovascular top 20 antihypertensive drugs in public hospitals in key cities in China from 2007 to 2016
The China Cardiovascular Disease Report 2016, compiled and released by the National Cardiovascular Disease Center, is the 11th annual report issued by China, which reflects the high attention paid by the Chinese government to the rising trend of cardiovascular disease. It has become an important project in the era of great health to prevent and treat hypertension, dyslipidemia, diabetes and obesity syndrome with a positive attitude of the whole people, and to take active and effective measures to curb the growth of cardiovascular disease.
The management of hypertension has a history of more than half a century, and relatively speaking, effective management of hyperlipidemia is only a nationwide action in the past 10 years. Statins have become milestones in the cholesterol lowering and blood lipid regulating program.
At present, statins in the blood lipid lowering market mainly include atorvastatin, rosuvastatin, pivastatin, simvastatin, pravastatin, fluvastatin, lovastatin, and more than 10 varieties of compound preparations, such as simvastatin+ezetimibe, atorvastatin+amlodipine, lovastatin+niacin.
Atorvastatin is the first drug in the blood lipid lowering market, and also the top three drugs in the cardiovascular market. According to CMH data of China Medical Information System, atorvastatin shows a rapid growth trend. In 2016, the sales of atorvastatin in domestic urban public hospitals were 6.075 billion yuan, the domestic retail pharmacy atorvastatin market had reached 2.431 billion yuan, and the two major mainstream terminals reached 8.506 billion yuan. The sales of atorvastatin in the six major markets of domestic urban public hospitals, county-level public hospitals, urban community medical care, township hospitals, physical pharmacies and online stores have exceeded 13 billion yuan.
According to HDM system data, in 2016, atorvastatin, rosuvastatin and pivastatin in public hospitals in key cities were among the top 3 drugs in the blood lipid lowering market, with year-on-year growth of 8.78%, 10.91% and 50.37% respectively. The six markets of the three major domestic terminals reached 23.827 billion yuan, and it is expected to reach 25 billion yuan in 2017.
Clopidogrel tops the market in heart medicine
Betrexaban, a new drug for cardiovascular and cerebrovascular systems, is another oral factor Xa inhibitor anticoagulant approved by Portola to be listed in the United States in the first half of 2017, and is the fourth drug with the same mechanism to be listed globally after rivalsaban, apixaban, and eduxaban. Betrexaban has the advantage of being an alternative to warfarin sodium, providing a new option for anticoagulation treatment of atrial fibrillation.
In the field of anticoagulant therapy for atrial fibrillation, with the introduction of rivalsaban, apixaban, dabigatran ester, tegrenol and prasugrel into clinical practice, the market for antithrombotic and anticoagulant therapy has been further improved. However, clopidogrel and low-molecular-weight heparin, which are mainstays in the Chinese market, still play an immeasurable role.
At the end of the 1990s, the US FDA approved Clopidogrel developed by Bristol Myers Squibb (BMS) to prevent and treat atherosclerotic diseases, myocardial infarction, stroke, peripheral vascular diseases and other diseases, becoming a landmark variety in the field of cardiovascular medicine.
Clopidogrel and aspirin small dose enteric coated tablets are known as the gold standard treatment plan for anti platelet aggregation and prevention of thrombosis, and are drugs to prevent myocardial infarction and stroke in the treatment of hypertension. Clopidogrel is jointly sold by Bristol Myers Squibb and Sanofi Aventis under the trade name Plavix. Clopidogrel is one of the earliest varieties developed and marketed in China. At present, 16 Clopidogrel API production lines have been built in China. In 2000, China first approved the registration and production of clopidogrel by Shenzhen Xinlitai Pharmaceutical, with the trade name of "Taijia".
Clopidogrel from Sanofi Aventis is marketed as "PLAVIX" in China. Driven by the original research drug and domestic drug, Clopidogrel has become a rapidly growing variety. The compound annual growth rate was 29.8% in the 10 years from 2005 to 2015. After the market developed to the platform stage, the growth slowed down in the past three years, and the consumption continued to rise.
HDM system data shows that in 2016, the market of clopidogrel in public hospitals in 16 key cities in China was 1.529 billion yuan, up 6.25% year on year. Among them, Sanofi Aventis's PLAVIX accounts for 58.84%, Shenzhen Xinlitai's Taijia accounts for 35.27%, and Lepu Pharmaceutical's Shuaike accounts for 5.89%. The growth rates of PLAVIX and TEGA were 5.24% and 3.66%, and the growth rate of Shueke of Lepu Pharmaceutical was 40.30%. In 2016, the overall sales scale of clopidogrel tablets in China exceeded 10 billion yuan, ranking first in the domestic heart drug market.
Figure 5 Clopidogrel market of public hospitals in key cities in China from 2007 to 2016
7 Antihypertensive drugs rank among the top 20 cardiovascular and cerebrovascular drugs
Calcium antagonists, renin angiotensin receptor antagonists and β Receptor blockers are a tripod of drugs in the antihypertensive market in China. Its mechanism of action is not only to control blood pressure, but also to protect the heart, brain, kidney and other important organs of hypertensive patients. In clinical treatment, it has positive effects on coronary heart disease, heart failure, arrhythmia, protection of renal function, reversal of left ventricular hypertrophy and vascular remodeling.
CMH data shows that in 2016, the market size of antihypertensive drugs in urban public hospitals in China was 36.6 billion yuan, an increase of 13% over the previous year. With the improvement of the medical security system, the market pattern shows that the top five calcium antagonists account for 34%, and renin angiotensin receptor antagonists account for 28%, β Blockers accounted for 8%, compound preparations for 7%, diuretics for 2%; Others accounted for 21%.
Among the top 20 varieties in the cardiovascular and cerebrovascular drug market of public hospitals in key cities, 7 drugs, including antihypertensive drugs amlodipine, levamlodipine, nifedipine, valsartan and compound, irbesartan and compound, losartan and compound, metoprolol, accounted for 21.40%.
Figure 6 Market performance of cardiovascular and cerebrovascular top 20 antihypertensive drugs in public hospitals in key cities in China from 2007 to 2016
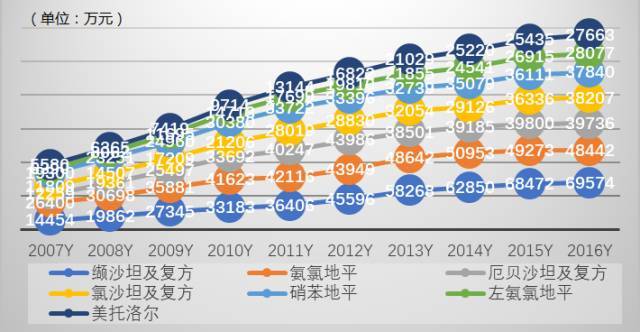
HDM system data shows that in 2016, the amount of calcium antagonists amlodipine, nifedipine and levamlodipine used in public hospitals in key cities was 484 million yuan, 378 million yuan and 281 million yuan respectively, an increase of 0.16%, 4.79% and 4.32% over the previous year; The amount of drugs used for the renin angiotensin receptor antagonist valsartan series, irbesartan series and losartan series was 696 million yuan, 397 million yuan and 382 million yuan respectively, an increase of 1.57%, -0.16% and 5.15% over the previous year. As a batch of new antihypertensive drugs enter the medical insurance category B, it will directly promote market growth.
key word: Top 20 accounts for more than 75%! Take a closer look at the leading varieties in the 100 billion level cardiovascular and cerebrovascular drug market







